IUCN/SSC Otter Specialist Group Bulletin

|
©IUCN/SCC Otter Specialist Group Citation: Marmontel, M., Isis Buck Silva, C., Botero-Arias, R. and Miguel, H.A. (2011) Rescue, Tagging and Release of a Neotropical Otter (Lontra longicaudis) in Western Brazilian Amazon. Proceedings of XIth International Otter Colloquium, IUCN Otter Spec. Group Bull. 28B: 36 - 46 Rescue, Tagging and Release of a Neotropical Otter (Lontra longicaudis) in Western Brazilian Amazon Miriam Marmontel1, Cristina Isis Buck Silva2, Robinson Botero-Arias1 and Hadad A. Miguel3
11Instituto de Desenvolvimento Sustentável Mamirauá, Estrada do Bexiga 2584, Bairro Fonte Boa, 69470-000 Tefé AM, Brazil. Email:
marmontel@mamiraua.org.br
|
 |
| (Received 8th October 2011, accepted 25th February 2012) |
|
Abstract: We report the rescue of a young Neotropical otter in western Brazilian Amazon and the process of quarantine, rehabilitation and preparation for release, including infectious disease tests. Prior to release a VHF transmitter was implanted in the intraperitoneal cavity of the animal. The otter was released in a sustainable development reserve in the vicinity of capture site, and had its movements followed for a short period. This is the first case of tagging and release of a Neotropical otter in the Amazon. |
| Keywords: Lontra longicaudis, conservation, mark-release |
| Française | Español | Português |
INTRODUCTION
Neotropical otters Lontra longicaudis (van Zyll de Jong, 1972)have a wide distribution throughout Latin America, occurring from northern Argentina to northwestern Mexico ( Chehébar, 1990 ; Redford and Eisenberg, 1992 ), both in deciduous and evergreen forests, in warm and cool climates ( Emmons, 1990 ). They are widely distributed within Brazil as well, from the Amazon and Pantanal to coastal rivers in the North and coastal and inland waters from Rio de Janeiro to Rio Grande do Sul ( Rosas, 2004 ).
Clear, fast-flowing rivers and streams, with abundant riparian vegetation are preferred ( Emmons, 1990 ; Redford and Eisenberg, 1999 ), but brackish and saltwater environments may also be used ( Blacher, 1987 ). The species feeds mainly on fish, especially from families Cichlidae, Anastomidae, Characidae and Pimelodidae ( Passamani and Camargo, 1995 ). Foraging takes place during the day, with emphasis in mid- to late afternoon ( Parera, 1993 ). Usually solitary, Neotropical otters pair up for breeding and female and 1-2 young are occasionally seen ( Parera, 1993 ). Cubbing may occur in nests built out of grass and leaves, or in hollow trees ( Harris, 1968 ; Eisenberg, 1989 ). Logs are also used for sprainting ( Macdonald and Mason, 1992 ; Parera, 1993 ). It is not a conspicuous species and in areas with higher human population, it is considered crepuscular, so most studies have been limited to analyzing signs (such as tracks, scats).
The Neotropical otter was heavily hunted for fur between 1950 and 1970, which resulted in local extinction over portions of the original range ( Brack-Egg, 1978 ; Donadio, 1978 ). It is currently protected throughout most of its range and listed under Appendix I of CITES ( CITES, 2009 ). The species is considered “data deficient” according to IUCN criteria ( Waldemarin and Alvares, 2008 ) and the Brazilian Action Plan for Aquatic Mammals ( IBAMA, 2001 ). Present population status is unknown. Few ecological studies on Neotropical river otters have been conducted in Brazil ( Pardini, 1996 ; Waldemarin, 1997 ; Quadros 1998 , Kasper et al., 2004 ), all in southern states.
The typical streamlined mustelid body form prevents the adaptation of radio-collars, but attempts at surgical implantation of transmitters have been successful with different species ( Melquist and Hornocker 1979 , 1983 ; Garshelis and Siniff 1983 ; Eagle et al. 1984 ; Hoover, 1984 ; Reid et al. 1986 ; Ralls et al. 1989; Ruiz-Olmo et al. 1991; Monnett and Rotterman 1995 ; Hernandez-Divers et al. 2001 ; Ó Néill et al. 2008 ). Advantages of this technique include the fact that the transmitter appears to be tolerated by the otter, and is not easily damaged or removed by the subject, and therefore it is suitable for long-term studies ( Garshelis and Siniff 1983 ). A telemetry device has been used once previously in a Neotropical otter in Brazil, again in the south. This paper describes the first attempt to follow and monitor a Neotropical otter through telemetry in the Amazon.
ANIMAL, MATERIAL AND METHODS
On March 24, 2009, a Neotropical otter (Lontra longicaudis) was confiscated by the Brazilian environmental law enforcement agency IBAMA in the town of Tefé, AM. The animal was rescued when a local man offered it for sale for R$ 50,00 (approx. U$ 25.00) in the Juruá sector of town. The otter had originally been caught four months prior in the Mineroá channel (São Sebastião do Baú community, Nogueira district - Fig. 1 ), city of Alvarães. During the month immediately before rescue it shared quarters with non-vaccinated domestic dogs at a house in town. Morphometrics and biological sampling were conducted within a few days of arrival, and the animal underwent a quarantine period in order to assess its condition and the possibility of releasing it back into the wild. Prior to release the animal underwent a surgical procedure for implantation of an intraperitoneal transmitter. All manipulative procedures to the animal were done in the presence of a veterinarian. This type of device is commonly used in otters, and was judged the most appropriate, given that the otter´s extremely flexible body and investigative behavior prevent the use of collars, belts or other external tags.
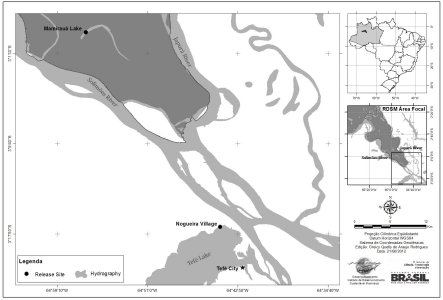 |
| Figure 1.
NLocation map showing rescue and release sites of Neotropical otter Click for larger image |
RESULTS
Exams and procedures
Upon rescue the animal was deemed to be in a general good nutritional condition, with no apparent wounds or defects. It was housed at a makeshift individual site with a cement floor (9.4 m2) and 2 m high walls within the IBAMA-Tefé facility. It was provided with a truck tire for shelter, a 500 l water tank with ladder and a 2nd, 500 l water tank, plus an artificial sand beach. Small, fresh, often live, fish (mostly sardines) were offered as food 2x day-1 (average 200 g day-1).
First inspection showed it to be a male of approximately 3 kg in weight. Due to the captivity period and contact with domestic animals, deworming was performed on April 4, 2009. Additional preparatory steps planned included haemogram, fecal parasitological exam, and tests for detection of infection by canine parvovirosis and distemper. Physical restraint was experimented with a noose, net or butterfly net, with best results with the latter.
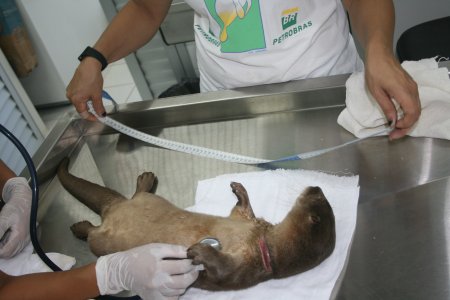
Further examination showed that the animal had a cotton rope around its neck which was digging into its muscle; so on April 11, 2009, anesthesia was performed to allow cleaning and suturing of the wound, as well as a detailed clinical examination, morphometrics and biological sampling for genetics and clinical analyses.
Anesthesia was obtained by chemical restraint with a combination of xylazine and ketamine. Pre-anesthesia was obtained with zylazine 10%, using 0.2 ml subcutaneous (SC). After 15 min, 0.2 ml ketamine chloridrate 10% was administered SC. As the animal showed responsive reflexes to external stimuli, 0.2 ml ketamine IM and local anesthesia with lidocaine 2% were administered. An additional supplementation was done 20 min later with 0.3 ml ketamine IM to conclude the procedure.
Wound treatment: There were 2 cervical, bilateral, lacerated wounds, 7.5 cm on the left and 11 cm on the right, caused by the rope used to physically restrain the animal. The wounds were cleaned, with debridation and revival of borders to allow suturing. Wounds were sutured with 3.0 nylon line and simple isolated stitches. Sodium rifamycin spray and topic repellent were used to top the suture, and the animal was administered 0.35 ml SC long-term antibiotic (benzathyne penicillin and procaine, streptomycin) and 0.06 ml SC anti-inflammatory (ketoprofen).
The whole procedure lasted 2¼ h (09h30-11h45), with 2½h post-anesthesia monitoring, when the animal showed good recovery.
|
|
Figure 1.
NLocation map showing rescue and release sites of Neotropical otter Click for larger image |
Morphometrics were obtained on April 11 ( Table 1 ), while the animal was under anesthesia.
| Table 1: Morphometrics of the rescued Neotropical otter | |||||
|
|
|||||
|
|
|||||
| Total length (including tail) | 82 cm | ||||
| Tail length | 37.5 cm | ||||
| Thorax girth | 32.5 cm | ||||
| Length of hind foot (right) | 9.76 cm | ||||
| Weight | 3100 g | ||||
|
|
|||||
The animal was considered to be a juvenile, as its measurements corresponded to the lower end of the range for adult Neotropical otters ( Harris, 1968 ; Emmons, 1990 ; Redford and Eisemberg, 1992 ; Kruuk, 2006 ).
The clinical exam revealed a good nutritional status, normal mucosa coloration and hydration status, and bright and uniform pelage, with absence of ectoparasites. Health parameters at the beginning and end of surgical procedure ( Table 2 ).
| Table 2: Health parameters of the Neotropical otter during surgery | ||
|
|
||
| Parameter | Start | End |
|
|
||
| Rectal Temperature | 37.8° C | 38.1°C |
| Cardiac Rate | 100 bpm | 160 bpm |
| Respiratory Rate | 20 bpm | 30 bpm |
|
|
||
Blood was collected with a Vacutainer system from the cephalic vein of the right anterior limb. Samples collected at the time included: 5 ml total blood with EDTA for CBC and genetic analysis, and 2 ml total blood without anticoagulant for biochemistry.
Fresh feces were collected from the soil on April 14 for parasitological exam. On May 4th, conjunctival swabs for canine parvovirosis and distemper were collected.
CBC was performed under a Neubauer chamber. The red blood cell series was normal; the white blood cell series showed discrete anisocytosis, intense leucocytosis, neutrophilia with bias to the left, and moderate lymphocytosis, monocytosis and eosinophilia ( Table 3 ).
The Willis-Mollay flotation method was negative for nematode eggs and protozoan oocytes, and the modified Dennis-Stones & Swanson sedimentation method was negative for cestode and trematode eggs.
Canine parvovirosis and distemper were tested with cromatographic immunoassay kits, using conjunctival swabs. Both tests came out negative.
Even though commercial, rather than specific, kits were used, and our still limited knowledge about the species, the quarantine period was considered large enough to allow manifestation of any disease the animal might carry.
Transmitter Implantation
The transmitter was a 3-stage lithium battery 3.6 v 2.6 ah LPI 2124 (Wildlife Materials), with coiled antenna (no external parts) for implant, with the mortality option. Specifications were as follows: dimensions 4.4 x 3.8 x 1.8 cm, 49 g, pulse width 25 ms, pulse rate 55 ppm, peak current 11 ma, power output –20 to –28 dBm and estimated battery life of 371 days. Frequency of the transmitter was 173.362 MHz.
On July 27, 2009, the transmitter was successfully implanted, following a full anesthesia. The animal remained under observation for 11 days, which allowed for healing. No further chemical restraint was performed and stitches were not removed.
Release
Given the known origin of the animal, its good health status and age class, IBAMA and the Mamirauá Institute for Sustainable Development (MISD) agreed on releasing the animal within the Mamirauá Sustainable Development Reserve (MSDR). The Mamirauá Lake, within the Mamirauá sector, was selected as the release site, due to the abundance of fish prey species, records of Neotropical otters in the region (by tourists and guides) and due to its status as a protected area. Nogueira district (where the animal was originally captured) is located 25 km in a straight line from the SE corner of MSDR, probably a good indication that the young male otter could be released there. Consultation with geneticists (Cristine Trinca and Eduardo Eizirik, pers. comm.) from PUC-RS suggested a genetic similarity between the (few) populations or subpopulations evaluated so far in the Amazon region, with no considerable genetic differentiation. Therefore, it was assumed that in terms of ecoregion or zoogeographic province, release in the MSDR would not constitute a conservation problem.
The State Center for Protected Areas (Centro Estadual de Unidades de Conservação CEUC) agreed with and authorized the joint effort by MISD and IBAMA to release the animal within MSDR, and MISD signed a responsibility document for release and post-release monitoring. The release process followed recommendations on Instrução Normativa 179/08, which regulates wildlife releases in Brazil.
On August 4, the otter was well and the healing process well advanced, release was planned for the weekend.
The animal was taken to the reserve on the morning of August 8th, and released in a floodplain area, on the right margin of the Mamiraua lake headwaters (S 02º59’39.2’’ W 064º56’44.4”). Because water levels were still very high, no high land was available. The release process started onboard the aluminum boat, with the engine off, at 11h30. The door of the transport container was opened, but the animal did not go out spontaneously, so the box was slightly tilted towards the water. By 11h43 the animal felt comfortable enough to jump out into the water. Once in the water, the animal was sighted only once.
Tracking
Tracking the released animal was considered a fundamental portion of the project, since it is the first time that it has been done with a Neotropical otter in the Amazon. A test of the transmitter was performed before release, and tracking was initiated immediately upon release.
Through reception of the transmitter signal it was possible to identify a tendency to circular movement around the release site immediately after release. After 30 minutes the animal had moved some 80 m away from the site (estimate based on basic triangulation). Monitoring continued until 12h50 and any approximation was avoided. Tracking was resumed at 14h30, and after several trials the signal was detected at a spot approximately 40 m from the release site (S 02º59’38.2’’ W 064º 56’43.3). The position of the animal was calculated at 60 m from the point where the signal was detected. The otter was spotted at 17h16 at 02º59’8.1’’ W 064º 56’5.3” and its behavior was observed for approximately 20 min.
VHF-monitoring was performed daily since release, around the area where the animal was released. Tracking was conducted by a local assistant aboard an aluminum canoe fitted with a 15-HP outboard motor, using a TRX-1000S Wildlife Materials “pushed” to 2000, 173-174 MHz range, 3-element 173 – 3FB Yagi antenna and David Clarke model H7050 earphones.
On August 9, 08h00, it became very hard to detect the signal, which was very faint and intermittent. Despite the faint signal the animal was visually spotted. Monitoring went on for 80 min but it was clear that the animal was responding to the researchers’ presence. Observations and tracking were terminated at 10h30, and resumed at 14h00, but no signal was detected then.
As of the end of September, the animal had not been found again. As the water level was still high at the time of release, it is possible that the animal moved long distances. The fact that the transmitter was implanted intraperitoneally, without an exposed antenna, may also help explain the difficulty in finding the signal. The receiver presented problems and had to undergo repair, and tracking could not be resumed as planned due to logistical difficulties.
DISCUSSION AND CONCLUSION
The only previous attempt to tag a Neotropical otter in its range occurred in 2003, when researchers live-trapped a specimen in a mangrove area during a mammal study in southeast Brazil. Transmitter attachment was improvised by using 2 collars around the otter´s body. The animal was monitored for 35 days, until it managed to remove the collars ( Nakano-Oliveira et al., 2004 ).
Ours was the first attempt to rescue and radio-track a Neotropical otter with an implanted transmitter, in the Amazon. Although the rehabilitation process was a success, we acknowledge our inability to track the animal past the first few days, which may have been due to the otter having moved a long distance from the release site, or even that the animal died. In at least one previous study ( Ó Néill et al. 2008 ) failure to capture the transmitter signal was attributed to dispersal movements
The transmitter may have malfunctioned or not be the most appropriate for our situation, and we may require to look into other choices of transmitters and release schedule. Our knowledge of the biology of Neotropical otters in the wild is still incipient, and conservation measures require improvements in that area to achieve positive results.
Acknowledgements - We would like to thank the Secretary of Sustainable Development for the permit to release the otter in the MISD. We are in debt with Nerinho and his team at the landing base in Tefé, for providing fish daily for Lorivaldo, the otter.
REFERENCES
Blacher, C. (1987). Ocorrência e preservação de Lutra longicaudis (Mammalia: Mustelidae) no litoral de Santa Catarina. Boletim da FBCN, 22: 105-117.
Brack-Egg, A. (1978). Situación actual de las nutrias en el Peru. In: Duplaix, N. (Ed.). Otters: Proceedings of the First Working Meeting of the Otter Specialist Group, IUCN, Morges, Switzerland, pp. 76-84.
Chehébar, C. (1990). Action plan for Latin American Otters. In: Foster-Turley, P., Macdonald, S., Mason, C. (Eds.). Otters: an action plan for their conservation. Proceedings of the IUCN, Otter Specialist Group Meeting, Gland, Switzerland, pp. 64-73.
CITES
http://www.cites.org/eng/resources/species.html
downloaded 28 October 2009
Colares, E.P., Best, R.C. (1991). Blood parameters of Amazon otters (Lutra longicaudis, Pteronura brasiliensis) (Carnivora, Mustelidae).
Comp. Biochem. Physiol., 99A: 513-515.
Donadio, A. (1978). Some comments on otter trade and legislation in Colombia. In: Duplaix, N. (Ed.). Otters: Proceedings of the First Working Meeting of the Otter Specialist Group. IUCN, Morges, Switzerland, pp. 34-42.
Eagle, T.C., Norris,J.C., Kuechle, V.B. (1984). Implanting radio transmitters in mink and Franklin´s ground squirrels. Wildl. Soc. Bull. 12: 180-184.
Eisenberg, J.F. (1989). Mammals of the Neotropics, the Northern Neotropics: Panama, Columbia, Venezuela, Guyana, Suriname, French Guiana. The University of Chicago Press, Chicago, 1: 449 pp.
Emmons, L.H. (1990). Neotropical rainforest mammals: a field guide. The University of Chicago Press, Chicago, 281 pp.
Foster-Turley, P., Macdonald, S., Mason, C. (Eds). (1990). Otters: an action plan for their conservation. Proceedings of the IUCN, Otter Specialist Group Meeting, Gland, Switzerland, 126 pp.
Garshelis, D.L., D.B. Siniff (1983). Evaluation of radio-transmitter attachments for sea otters. Wild. Soc. Bull. 11(4): 378-383.
Harris, C.J. (1968). Otters: a study of the recent Lutrinae. Weinfield and Nicolson, London, 397 pp.
Hernandez-Divers, S.M., Kollias, G.V., Abou-Madi, N. (2001). Surgical technique for intra-abdomiinal radiotransmitter placement in North American river otters (Lontra canadensis).
J. Zoo and Wildlife Med.
32(2):202-205.
Hoover, J.P. (1984). Surgical implantation of radiotelemetry devices in American river otters.
JAVMA
185(11): 1317-1320.
IBAMA. (2001). Mamíferos aquáticos do Brasil - Plano de Ação, versão II, 2. ed. Brasília, Instituto Brasileiro de Meio Ambiente e dos Recursos Naturais Renováveis,10 pp.
Kasper, C.B., Feldens, M.J., Salvi, J., Grillo, H.C.Z. (2004). Estudo preliminar sobre a ecologia de Lontra longicaudis (Olfers) (Carnivora, Mustelidae) no Vale do Taquari, Sul do Brasil. Rev. Bras. Biol. 21(1): 65-72.
Kruuk, H. (2006). Otters - ecology, behavior and conservation. Oxford University Press, Oxford, 265 pp.
Larivière, S. (1999). Lontra longicaudis.
Mamm. Spec. 609: 1-5.
Macdonald, S., Mason, C. (1992). A note on Lutra longicaudis in Costa Rica.
IUCN/SSC Otter Spec. Group Bull. 7: 37-38.
Mason, C., Macdonald, S. (1990). Conclusions and priorities for otter conservation. In: Foster-Turley, P., Macdonald, S., Mason, C. (Eds.). Otters: an action plan for their conservation. IUCN Otter Spec. Group, Gland, Switzerland, pp. 80-88.
Melquist, W.E., Hornocker, M.G. (1979). Development and use of a telemetry technique for studying river otter. In: Long, F.M. (Ed.). Second Intl. Conf. Wildl. Biotelemetry. Laramie, Wyoming, pp. 104-114.
Melquist, W.E., Hornocker, M.G. (1983.) Ecology of river otters in West Central Idaho. Wildl. Monogr. 83: 3-60.
Monnet, C., Rotterman, L.M. (1995). Mortality and reproduction of female sea otters in Prince William Sound, Alaska.
Exxon Valdez Oil Spill State/Federal Natural Resource Damage Assessment Final Report, Marine Mammal Study 6-13, Final Report, Anchorage AK, iv + 12pp.
Nakano-Oliveira, E., Fusco, R., dos Santos, E.A.V., Monteiro-Filho, E.L.A. (2004). New information about the behavior of Lontra longicaudis (Carnívora: Mustelidae) by radio-telemetry.
IUCN/SSC Otter Spec. Group Bull. 21(1):31-35.
Ó Néill, L., Wilson, P, de Jongh, A., de Jong, T, Rochford, J. (2008). Field techniques for handling, anaesthetizing and fitting radio-transmitters to Eurasian otters (Lutra lutra). Eur. J. Wildl. Res. 54: 681-687.
Pardini, R. (1996). Estudo sobre a ecologia de Lontra longicaudis no Vale do Alto rio Ribeira, Iporanga - SP (Carnivora: Mustelidade). MSc thesis, Universidade de São Paulo, Instituto de Biociências, São Paulo.
Parera, A. (1993). The neotropical river otter Lutra longicaudis in Iberá lagoon, Argentina.
IUCN/SSC Otter Spec. Group Bull. 8: 13-16.
Passamani, M., Camargo, S.L. (1995). Diet of the river otter Lutra longicaudis in Furnas reservoir, south-eastern Brazil.
IUCN/SSC Otter Spec. Group Bull., 12: 32-33.
Quadros, J. (1998). Aspectos da ecologia de Lontra longicaudis (Olfers, 1818) em uma área de Floresta Atlântica de Planície, Município de Itapoá-SC. MSc thesis, Universidade Federal do Paraná, Setor de Ciências Biológicas, Curitiba.
Ralls, K., Siniff, D.B., Williams, T.D., Kuechle, V.B. (1989). An intraperitoneal radio transmitter for sea otters.
Mar. Mam. Sci.
5(4): 376-381.
Redford, K.H., Eisenberg, J.F. (1992). Mammals of the Neotropics: the Southern Cone. Chile, Argentina, Uruguay, Paraguay. The University of Chicago Press, Chicago, 2 : 430 pp.
Reid, D.G., Melquist, W.E., Woolington, J.D., Noll, J.M. (1986). Reproductive effects of intraperitoneal transmitter implants in river otters. J. Wildl. Manage. 50(1):92-94.
Rosas, F.C.W. (2004). Lontra, Lontra longicaudis (Carnívora: Mustelidae). In: Cintra, R. (Coord.). História natural, ecologia e conservação de algumas espécies de plantas e animais da Amazônia. EDUA/INPA/FAPEAM. (série: Biblioteca Científica da Amazônia), Manaus, pp. 261-264.
Ruiz-Olmo, J., Jimenez, J., Marco, I. (1991). Radiotracking a translocated otter in Spain.
IUCN/SSC Otter Spec. Group Bull. 6: 6-7.
Silveira, L., Furtado, M.M., Rosas, F.C.W., Silva, L.C.L.C., Cabral, M.M.M., Tôrres, N.M., Sollmann, R., Kouba, A., Jácomo, A.T.A. (2011). Tagging giant otters (Pteronura brasiliensis) (Carnivora, Mustelidae) for radio-telemetry studies. Aquatic Mammals
37(2): 208-212.
Waldemarin, H.F. (1997). Estudo da ecologia de lontras (Lontra longicaudis) no Parque Nacional da Lagoa do Peixe: manejo e conservação. Monografia. Fundação Universidade do Rio Grande, Rio Grande.
Waldemarin, H.F., Alvares, R. (2008). Lontra longicaudis. In: IUCN 2009. IUCN Red List of Threatened Species. Version 2009.1. <
www.iucnredlist.org
>. Downloaded on 28 October 2009.
Résumé : Sauvetage, Marquage et Relacher d’une Loutre à Longue Queue (Lontra longicaudis) sur Amazone Occidental du Bresil
Nous rapportons ici le sauvetage d'une jeune Loutre à longue queue dans l'ouest de l'Amazonie brésilienne, le processus de mise en quarantaine, sa réhabilitation, sa préparation à la libération mais aussi les analyses de maladies infectieuses. Avant le relâcher, un émetteur VHF a été implanté dans la cavité intrapéritonéale de l'animal. La loutre a été libérée dans une réserve de développement durable dans le voisinage du site de capture et ses mouvements ont été suivis pendant une courte période. C'est le premier cas de marquage et de libération d'une Loutre à longue queue dans l’Amazone.
Revenez au dessus
Resumen: Rescate, Marcaje y Liberación de una Nutria Neotropical (Lontra longicaudis) en la Amazonia Brasileña Occidental
Reportamos el rescate de un juvenil de Nutria Neotropical en la Amazonia brasileña occidental y el proceso de cuarentena, rehabilitación y preparación para su liberación, incluyendo pruebas de enfermedades infecciosas. Previo a la liberación se le implanto un transmisor de VHF en la cavidad intraperitoneal del animal. La nutria fue liberada en una reserva de desarrollo sustentable en las inmediaciones del sitio de captura, y se siguieron sus movimientos durante un corto período. Este es el primer caso de etiquetado y liberación de una nutria Neotropical en el Amazonas.
Vuelva a la tapa
Resumo: Rescate, Marcage y Soltura d una Nutria Neotropical (Lontra longicaudis) en la Amazonia Occidental Brasileña
Se describe el rescate de uma nutria neotropical juvenil en la Amazonia occidental brasileña, y el proceso de cuarentena, rehabilitación y preparación para soltura, incluyendo testes para enfermedades infecciosas. Ante la soltura se implantó un transmisor VHF en la cavidad intraperitoneal del animal. La nutria fue liberada en una reserva de desarrollo sostenible en las proximidades del lugar de captura, y sus movimientos fueran monitoreados por un corto periodo de tiempo. Ese es el primer caso de marcage y soltura de una nutria neotropical en la Amazonia.
Voltar ao topo