IUCN/SSC Otter Specialist Group Bulletin

|
©IUCN/SCC Otter Specialist Group Volume 30 Issue 1 (January 2013) Citation: Hussain, S.A. (2013). Activity Pattern, Behavioural Activity and Interspecific Interaction of Smooth-Coated Otter (Lutrogale perspicillata) in National Chambal Sanctuary, India IUCN Otter Spec. Group Bull. 30 (1): 5 - 17 Activity Pattern, Behavioural Activity and Interspecific Interaction of Smooth-Coated Otter (Lutrogale perspicillata) in National Chambal Sanctuary, India Syed Ainul Hussain 1
1Wildlife Institute of India, Post Box # 18, Dehra Dun -248001, India . e-mail:
hussain@wii.gov.in
|
 |
| (Received 17th April 2012, accepted 22nd October 2012) |
| Abstract: The activity pattern, behavioural activity and interspecific interactions of smooth-coated otter (Lutrogale perspicillata) with crocodilians viz. gharial (Gavialis gangeticus) and mugger (Crocodylus palustris) was examined by monitoring four radio-implanted otters in the National Chambal Sanctuary, India. Based on the percentage of active telemetry recordings, the observed overall activity patterns of otters were in the form of a bimodal curve in which two periods of high activity were separated by a period of relative inactivity. In summer, the relative period of inactivity was greater than the winter and monsoons. Otters were more diurnal during winter than in summer or monsoon. Travelling and grooming were the most prevalent activities recorded during 422 sightings. Though the habitats and the diet of otters overlapped with gharial and mugger, their diurnal activity pattern was different and spatially they maintained a mutually agreeable strategy to avoid conflicts in sharing resources for basking and grooming. |
| Keywords: smooth-coated otter; Lutrogale perspicillata; behavior; activity pattern; interspecific interactions; Gavialis gangeticus; Crocodylus palustris |
| Française | Español |
INTRODUCTION
Otters being the topmost predators of the aquatic systems, its presence indicate the health of the wetland ecosystems. The relationship of otters and their prey appears to be similar to most other carnivores, but otters have to bear unusual costs for their foraging trips in the huge energetic requirements of keeping warm while fishing. This implies that otters will be able to sustain themselves only if they are able to consume their food quickly with the least foraging expense, which would otherwise be extremely high (Kruuk, 1995). Synchrony of activity phasing between predators and prey has been found in many carnivores. For example, seasonal foraging patterns in spotted-necked otter reflected a correlation between activity of the prey and hunting success of the otters (Perrin and Carranza, 2000). Food resource abundance and dispersion affecting foraging patterns and social behaviour has also been reported for several carnivores (Schaller, 1972, Kruuk and Hewson, 1978; Macdonald and Mason, 1983; Bekoff et al., 1984; Mills, 1989).
The smooth-coated otter (Lutrogale perspicillata, Geoffroy) ( Figure 1 ) is distributed throughout southern Asia from Indonesia, through southeast Asia, and westwards through southern China and India, with an isolated population L. p. maxwelli in the marshes of Iraq (Mason and Macdonald, 1986; Foster-Turley and Santiapillai, 1990; Hussain et al., 2008). On the Indian subcontinent, the smooth-coated otter is distributed from the foothills of Himalayas southward to southern India ( Pocock, 1949 ; Prater, 1971 , Hussain 1993 ; Hussain and Choudhury, 1997 ; Hussain, 2002 ; Nawab and Hussain, 2012a ) occurring along the major rivers with other aquatic carnivores such as crocodiles ( Prater, 1971 ). The smooth-coated otter is a large otter, weighing around 8-11 kg, distributed throughout South and Southeast Asia. It is a semi-aquatic social carnivore found in large groups ( Foster-Turley, 1992 ; Hussain, 1993 ; 1996 ), with the basic family group consisting of an adult female and her offspring, the father and older siblings often joining the group ( Lekagul and McNeely, 1988 ; Hussain, 1996 ). It is considered an endangered species and is listed in Schedule I of the Indian Wildlife (Protection) Act, 1972, “Vulnerable” in IUCN Red List ( Hussain et al., 2008 ) and Appendix II of CITES. In spite of its wide distribution, very few studies address the ecology and behaviour of smooth-coated otter. Because of the limited knowledge about these otters, especially the Asian species, practical conservation measures have not been developed. Therefore, the Wildlife Institute of India undertook a project to study the ecology of smooth-coated otters in National Chambal Sanctuary. This paper describes the activity pattern, behavioural activities and its interaction with crocodilians along the Chambal River in National Chambal Sanctuary, India.
 |
|
Figure 1.
Smooth-coated otter with cubs (Photo credit: Bivash Pandav) (click for larger version) |
STUDY AREA
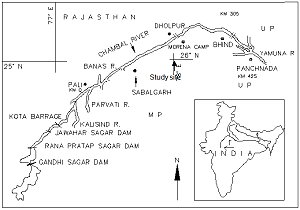
The Chambal is a clear and fast flowing river that originates from the Singar Chouri peak in the Vindhya Range, central India. Lying between 24°55’–26°50’N and 75°34’–79°18’E, it flows northeast and joins the Yamuna River to form the greater Gangetic drainage system ( Figure 2 ). A 600 km stretch of the Chambal river has been protected as the National Chambal Sanctuary for the conservation and management of gharial (Gavialis gangeticus, Crocodylia) ( Hussain, 1990 ; 1999 ; 2009 ). The area lies within the semi-arid zone of northwestern India at the border of Madhya Pradesh and Rajasthan states. The temperature of the area ranges from 20°C to 46°C. The annual precipitation largely depends on the southwestern monsoon which lasts from the third week of June until late September with 500–600 mm of rainfall. The natural vegetation of much of the Sanctuary area is ravine thorn forest ( Champion and Seth, 1968 ). Evergreen riparian vegetation is completely absent. The severely eroded river banks and adjacent ravine lands have sparse ground cover ( Hussain, 1993 ). Apart from the gharial ( Figure 6b )and smooth-coated otter, the fauna of the Sanctuary includes the marsh crocodile or mugger (Crocodylus palustris, Figure 6a ), seven species of freshwater turtles, Ganges river dolphin (Platanista gangetica),and 78 species of wetland birds ( Hussain, 1993 ). The intensive study area lies along a 17 km stretch of the Chambal River ( Figure 2 ). In this stretch the habitat consisted of rocky, clay, and sandy river banks often interspersed with marsh, dominated by Typha, and bankside vegetation, dominated by Tamarix spp. ( Hussain, 1993 ; Hussain and Choudhury, 1995 ).
 |
|
Figure 2.
Smooth-coated otter with cubs (Photo credit: Bivash Pandav) (click for larger version) |
Unlike other rivers of the greater Ganges drainage system, the Chambal River is relatively unpolluted ( Hussain and Singh, 1999 ). The water quality exhibited very low suspended solids with low biological oxygen demand (BOD) and high dissolved oxygen (DO). There was no indication of organic matter discharge or eutrophication in the river as the value of chemical oxygen demand (COD), ammonia (NH4), and phosphate (PO4) were below the detection limits. The essential cations (Ca, Mg, Na, and K) were also within the range to support aquatic organisms. On the basis of standards set by the Central Pollution Control Board, Government of India, the Chambal River water can be considered as `A' category. By comparing the water quality parameter with ranges given by Allen (1989) , the Chambal River is considered clean.
METHODSDuring February 1990 and March 1992, I captured and monitored the activity of four smooth-coated otters in National Chambal Sanctuary. I used Tomahawk live traps and leghold traps to capture the study animals. I immobilized them with a combination of ketamine and xylazine hydrochloride ( Hussain et al., 1996 ) and surgically implanted them with radio-transmitters (Model - IMP 400 VHF; Telonics, Arizona, USA). After holding the otters in a squeeze cage to ensure their surgical sites were properly healed, I released them at the capture sites, five to six days after surgery. I monitored activity of the otters for a period of 11 to 22 months ( Table 1 ). On the basis of size and skin texture, the captured otters were classified into juveniles, sub adults, and adults ( Table 1 ). I radio-tracked the otters once daily and recorded their locations on a 1:50,000-scale map with 250-m grids laid over the river. Activity of instrumented otters was recorded based on changes in signal volume and by direct observations. The intensity of the signal was regular when the animal was resting and irregular when it was moving ( Voight and Tinline, 1979 ). I obtained 1048 radio-locations and 422 sightings of the otters during a period of 24 months (for details see Hussain and Choudhury, 1995 ).
I determined diurnal activities such as foraging, feeding, travelling, and social behaviour by visual observations of instrumented and non-instrumented otters. A 24-hour monitoring effort was conducted once in every two weeks with otter locations and activity recorded at one-hour intervals.
The following criteria were used to classify activity and behavior:
- Telemetry
- Active : Any type of movement leading to a change in signal direction.
- Inactive: No change in signal volume or direction.
- Visual observations
- Foraging: Searching for and consuming food.
- Travelling: Intentional movement from one location to another.
- Grooming: Rolling on sand or other related activities.
- Marking: Defecating, urinating, or inspecting such sites.
- Resting: Sleeping at day time.
- Social: Any type of behavioural interaction between two or more individuals.
- Others: Any type of physical activity (play, territorial, digging).
During the radio monitoring period whenever otters were seen, I recorded the kind of river bank nearest to sighting locations, number of otters seen, and their distance from other aquatic fauna if present. At each sighting, a note on the behaviour of the other aquatic fauna also was made. I also monitored the basking behaviour of crocodilians to document peak haul-out and retreat times. Nests of crocodilians and turtles within the home range of otters at the radio-tracking site were monitored to see if they were disturbed by otters.
During the study, I collected 553 spraint samples from a 195 km stretch of the Sanctuary. Collections were made from the entrance of dens, communal sprainting sites adjacent to dens, and feeding areas. Only one sample was collected from each site. Attempts were made to collect fresh spraints, but during the monsoon all spraints were collected. I recorded date and location for each spraint at the time of collection and sun dried the samples, which were stored in paper bags, at the camp. The samples were later soaked in water and mixed with a detergent for 5-6 hours. After washing in running tap water over a 1-mm sieve, the cleaned samples were dried in shade and kept in plastic bags for sorting of prey remains. I sorted prey remains from cleaned spraints by using a hand lens or a compound microscope. All remains were identified by comparison with references of prey parts made during the study. Effort was made to identify the prey to species level but this was not always possible, hence some prey remains were identified only to genus level. Similarly, 25 scat samples of mugger (Crocodylus palustris) were collected from the study site during November 1989 to April 1990 and analysed using similar methods.
RESULTS
Activity patterns
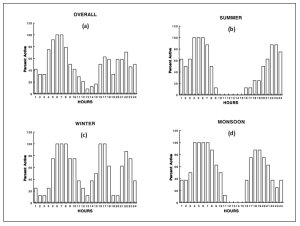
After release, all the instrumented otters remained together during the radio-tracking period, thus the activities recorded were group activities. Based on the percentage of active telemetry recordings, the annual activity pattern of smooth-coated otters in the National Chambal Sanctuary was in the form of a bimodal curve, in which two periods of high activity were separated by a period of relative inactivity. The first active period started after midnight and continued until late morning (10:00-11:00 hours), then there was a second active period which continued from approximately 16:00-17:00 hrs until 01:00 hrs (Figure 3a ). The period when inactivity was most distinct was during 09:00–17:00 hours during summer ( Figure 3b ). On a seasonal basis, activity patterns during winter were different from those of summer or monsoon. In winter the pattern of activity was trimodal, with the peak activity occurring between 06:00 to 08:00, 16:00 to 17:00 and 21:00 to 23:00 hours ( Figure 3d ). Difference was also found between daytime and nighttime activities in different seasons ( Figure 3b,c,d ). This indicated that the otters were nocturnal in their behaviour. Otters were more diurnal during winter than in the summer or monsoon. Seasonally the smooth-coated otter were more active during winter (36.1%) and progressively less active in summer (29.2%) or monsoon (34.7%).
 |
|
Figure 3.
Activity patterns of radio-tagged smooth-coated otters in National Chambal Sanctuary, India, 1990–1992. (click for larger version) |
Behavioural activities
Behavioural activity was recorded during 422 sightings made at the time of radio-tracking the otters. Of the nine types of behaviours I identified, more than one type was observed during each sighting ( Table 2 ). Thus, I recorded 561 behavioural activities during the study ( Table 2 ). These activities were observed during different months and at different times of the day. Travelling was recorded in 46% of the visual observations and it constituted 34.6% of the otters’ activity. Travelling was often on land. Most travels were between den and foraging sites. Foraging was recorded during 20% of the visual observations period. Most of the foraging activities involved either probing along the water edge, singly or in groups, or along rapids, moving upstream in semi-circles. Twice otters were observed attempting to catch blue rock pigeon (Columba livia) on land.
Most of the grooming behaviour was self grooming. This consisted of rolling on sand for five to fifteen minutes with intermittent dips in water and drying by rolling. Grooming was observed 147 times and constituted 35% of the total sightings. It was the second most prevalent activity of otters. Marking by defecation and urination or otherwise occurred mostly adjacent to dens, at grooming sites, along the usual travel routes and near the foraging sites ( Figure 4 ). Marking often occurred in groups while otters were grooming or foraging. It appeared that marking by one individual induced this behaviour among other members of the group. Males sniffed and examined the area before marking. Resting behaviour was observed mostly during winter. Otters were seen sleeping on sand or on rocks adjacent to the entrance of dens or were lying idle and often gazing into the water. Play behaviour comprised 4.5% percent of the sightings. This included juveniles chasing each other on land or in water and wrestling while the rest of the group was engaged in self grooming. Of the seven observations of social behaviours (except foraging), three involved transferring young ones from one den to another; and the remainder involved sniffing each other when two or more otters of same size met. Digging was observed during evening hours in the month of November ( Table 2 ). Digging was observed to be done by one otter of the group while the rest were engaged in auto grooming. Evidence of digging silt from natural caves was detected in October, which is the littering season.
 |
|
Figure 4. Marking smooth-coated otter (Photo credit: Anoop, K.R.). (click for larger version) |
Dietary overlap of smooth-coated otter and marsh crocodile
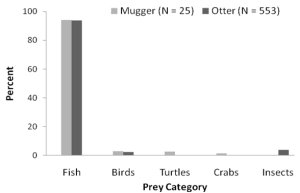
The smooth-coated otter is primarily a fish eater ( Hussain and Choudhury, 1998 ; Nawab and Hussain, 2012b ). In this study the relative frequency of occurrence of prey remains in otter spraints was 93.8% fish, 3.8% invertebrates, and 2.4% amphibians and birds ( Figure 5 ). Three percent of fish remains and 0.9% of birds could not be identified from the spraints. Apart from these, 46 occurrences of molluscs, 12 of vegetative matter (grassroots or grass) and seven occurrences of mammalian hair were also recorded. All the mammalian hairs were identified as otter hair, which might have been ingested by the otters while grooming. Mollusc shells and vegetative matter were ingested as secondary items, which were later confirmed by analysing gut contents of a major prey Rita rita. It was not clear whether the insects consumed were direct or due to secondary ingestion. Analysis of mugger droppings revealed that 94% of the marsh crocodile’s diet consisted of fish, 3% birds, 2.6% turtles and 1.5% invertebrates like crab and insects ( Figure 5 ). Of the fish diet, 74% was composed of unidentified Cyprinidae, 22% unidentified Bagaridae and 4% unidentified fish. The gharial is reported to be an exclusive fish eater ( Bustard, 1984 ), preferring lesser carps. Along the Chambal River, gharials were observed eating cat fish (Mystus spp.) during field work.
 |
|
Figure 5. Marking smooth-coated otter (Photo credit: Anoop, K.R.). (click for larger version) |
Spatio-temporal relationship between smooth-coated otter and other species
Interspecific interaction was the most important behaviour observed in the resident adult male, which chased a large gharial (2.5–3.0 m) on two occasions. Apart from this, yearlings were seen chasing two basking turtles (Kachuga kachuga) from their regular grooming sites. The number of gharial and marsh crocodile seen during the study period at the radio-tracking sites between km 160 to 175 is presented in Table 3 . Both the gharial and mugger populations in the radio-tracking site were found to be increasing at the rate of 4% percent per annum ( Hussain, 1990 ; 1999 ). Although breeding was recorded, no increase in the group size of otters or in the number of otter dens was recorded from this site. During the intensive monitoring of otters using radio-telemetry, 80% of the gharial sightings were on sandy banks/substrata and 19% were on rocky banks/substrata. In contrast, 68.9% of the sightings of mugger was on rocky banks/substrata and 10.4% and 6.7% on clay banks/substrata and sandy banks/substrata, respectively. Sandy and rocky substratums were used extensively by otters for grooming and marking. However, only 2.6% of the radiolocations for tagged otters were observed on sandy banks, while 77.9% were observed on rocky banks and 19% were on river bank with dense vegetation.
a
 |
b
 |
| Figure 6. Mugger or Indian marsh crocodile (a) and gharail (b) in National Chambal Sanctuary, India (click for larger version) |
DISCUSSION
Many animal species exhibit an endogenous rhythm of activity that approximates a diel cycle or twenty-four hour period ( Harker, 1964 ; Eisenberg, 1981 ). The question is whether such a rhythm is present in a semi-aquatic carnivore like otters, and if yes, what are the factors that influence it? Variations in the pattern of activity of carnivores is largely influenced by numerous factors such as periodicity resulting from physiological changes ( Sollberger, 1965 ), synchrony in diel activity between predator and their prey ( Melquist and Hornocker, 1983 ), climatic conditions of the area, the types of habitats in which they live, and the degree of human interference. By conducting a four year study on North American river otters Lontra canadensis, Melquist and Hornocker (1983) concluded that it was difficult to determine the role of innate physiological rhythms of river otters, because activity rhythms of river otters were influenced by numerous factors, any one of which may have had an overriding effect on others. However, in my study, I demonstrated the smooth-coated otters exhibited pronounced rhythmic activity all through the year, which was stronger in the summer and monsoon than in winter.
Some otters are thought to be nocturnal ( Macdonald and Mason, 1983 ; Chanin, 1985 ), while others may be diurnal (e.g. European otter on the Shetland coast, Kruuk, 1995 on the Scottish west coast; river otters in Idaho, Melquist and Hornocker, 1983 ). The nocturnal behaviour of smooth-coated otters in this study in a tropical, semi-arid environment of the Chambal River might be an adaptation for exploitation of prey, avoidance of disturbance, and limiting exposure to tropical heat. In this study the activity of fish was mostly restricted to the dark period (night)(personal observation). In American river otter in Idaho ( Melquiest and Hornocker, 1983 ) and in European otters along the Shetland coast ( Kruuk, 1995 ) this phenomenon was explained as that fish those are slow moving in their nature often hide under stones and weeds during day time, they are easy to catch by otters, then while they are active at night. Martin et al. (2010) reported similar observations for river otter in Canada, where otters were active throughout the day but with bimodal peaks during late evening and early morning hours. The spotted-necked otter (Lutra maculicollis) was found active when its efficiency in detecting its fish prey by sight was high, which was during daylight and moonlight in the Natal Drakensberg, South Africa ( Perrin and Carranza, 2000 ).
Apart from fish activity as a factor in otter activity, the tide also has an important effect on almost all coastal life including otters ( Kruuk, 1995 ). Because otters have a rapid gut-passage time ( Mason and Macdonald, 1986 ), one might expect periods of active foraging bouts at regular intervals. However, I did not observe this for smooth-coated otters. The otters were mostly inactive throughout the day in summer and monsoon. This indicates that apart from the availability of prey, other factors such as temperature and anthropogenic disturbances influence activity patterns.
Functional classification of behavioural elements is important to categorize behaviour into physiologically related groups (
Eisenberg, 1981
). In the case of otters, if this sequence of behavioural acts are identified and grouped, this will help in understanding behavioural responses by semi-aquatic carnivores to external stimuli. From the behavioural activity I recorded during this telemetric study, it was difficult to conclude the importance of physiologically-related activity to otters because there was sighting bias due to nocturnal behaviour and the secretive nature of the species. Travelling and grooming were the most prevalent activities recorded during 422 sightings. Grooming was recorded as the second important activity of otters because it plays an important role in its physiology. It increases insulation and blood circulation, decreases heat loss, and helps in maintaining general health of otters. In a study along the Shetland coast,
Nolet and Kruuk (1989)
found that, compared to sea otters, Eurasian otters spent up to 4.5 times less effort per day hunting but devoted the same time per day to grooming. Foraging was found to be the second prevalent activity but this again appears to be biased. Habitat and diet of smooth-coated otters overlapped with muggers; however no threat through interspecific competition was visualized. The degree of overlap in the utilization of fish may not be critical as fishing is banned in the Sanctuary. Interaction with other aquatic fauna such as crocodiles and turtles illustrate the behavioural flexibility, adaptive ability, and positive selective significance of this species.
Acknowledgements - This study was carried out through the project “Ecology of aquatic mammals in National Chambal Sanctuary” funded by the Wildlife Institute of India. I am extremely grateful to Director and Deans of the Institute for extending technical and logistic support. I am extremely grateful to two anonymous reviewers for their comments for improving the quality of this manuscript.
REFERENCES
Allen, S.E. (1989). Chemical analysis of ecological materials. Blackwell Scientific Publications, London.
Bekoff, M., Daniels, T.J., Gittleman, J.L. (1984). Life history patterns and the comparative social ecology of carnivores. Annual Review of Ecology, Evolution and Systematics, 15: 191-232.
Bustard, H.R. (1984). Breeding the Gharial (Gavialis gangeticus): Captive breeding a key conservation strategy for endangered crocodilians. Symp. Zool. Soc. London; 52: 385-406.
Champion, H.G., Seth, S.K. (1968). A revised survey of the forest types of India. Manager of Publication, Delhi. pp. 404.
Chanin, P. ( 1985) . The Natural History of Otters. New York: Facts on File.
Eisenberg, J.F. (1981). The mammalian radiation. An analysis of trends in evolution, adaptation, and behaviour. University of Chicago Press,Chicago.
Foster-Turley, P., Santiapillai, C. (1990). Action plan for Asian otters, In: Otters, an action plan for their conservation. Foster-Turley, P., Macdonald S., Mason, C.F. (Eds.) IUCN/SSC, Otter Specialist Group. IUCN, Gland.
Foster-Turley, P. (1992). Conservation ecology of sympatric Asian otters Aonyx cinerea and Lutra perspicillata, PhD thesis, University of Florida.
Harker, J. E. (1964).
The Physiology of Diurnal Rhythms. Cambridge University Press, Cambridge.
Hussain, S.A. (1990). Ecology of gharial in National Chambal Sanctuary. M Phil thesis, Centre for Wildlife and Ornithology, Aligarh Muslim University, Aligarh.
Hussain, S.A. (1993). Aspects of the ecology of smooth-coated otter (Lutra perspicillata) in National Chambal Sanctuary. PhD Thesis, Centre for Wildlife and Ornithology, Aligarh Muslim University, Aligarh.
Hussain, S.A. (1996). Group size, group structure and breeding in smooth-coated otter Lutra perspicillata, Geoffroy (Carnivora, Mustelidae) in National Chambal Sanctuary, India. Mammalia, 60 (2): 289-297.
Hussain, S.A.
(1999). Reproductive success, hatchling survival and growth of a managed population of Ganges gharial. Biological Conservation.
87:261-268.
Hussain, S.A. (2002). Conservation status of otters in the Tarai and Lower Himalayas of Uttar Pradesh, India. In Otter Conservation-An example for a sustainable use of wetlands. IUCN Otter Specialist Group Bulletin:
Proceedings from VIIth International Colloquium, Trebon, Czech Republic, 2002
19A: 131-142.
Hussain, S.A. (2009). Basking site and water depth selection by gharial Gavialis gangeticus in National Chambal Sanctuary and its implication for river conservation. Aquatic Conservation,19: 127-133.
Hussain, S.A., Choudhury, B.C. (1995). Seasonal movement, home range and habitat use by smooth-coated otters Lutra perspicillata in National Chambal Sanctuary, India. pp. 45-55. In: Proceedings of VI. International Otter Colloquium, Pietermaritzburg. Reuther, C., Rowe-Rowe, D. (Eds.). Aktion Fischotterschutz Hankensbüttel.
Hussain, S.A., Choudhury, B.C. (1997). Status and distribution of smooth-coated otters Lutra perspicillata in National Chambal Sanctuary. Biological Conservation, 80: 199-206.
Hussain, S.A., Choudhury, B.C. (1998). Feeding ecology of smooth-coated otter Lutra perspicillata in National Chambal Sanctuary. Proceedings of the Symposia of the Zoological Society of London, Behaviour and ecology of riparian mammals,
71: 229-250
Hussain, S.A., Singh, R.K. (1999). Ecological survey of the National Chambal Sanctuary to assess the need for desiltation. Study report. Wildlife Institute of India, Dehra Dun.
Hussain, S.A., Malik, P.K., Choudhury, B.C. (1996). Chemical immobilization of smooth-coated otter using a combination of ketamine and xylazine hydrochloride in National Chambal Sanctuary. Journal of Bombay Natural. History Society,
93(4): 214-218.
Hussain, S.A., de Silva, P.K., Mostafa, F.M. (2008).
Lutrogale perspicillata. In: 2008 IUCN Red List of Threatened Species.
http://www.iucnredlist.org/apps/redlist/details/12427/0
. Downloaded on 07 January 2012.
Kruuk, H. (1995). Wildotters - Predation and populations. Oxford University Press.
Kruuk, H., Hewson, R. (1978). Spacing and foraging of otters (Lutra lutra) in a marine habitat. J. Zool.,London. 185: 205-212.
Lekagul, B., McNeely, J.A. (1988).
Mammals of Thailand, 2nd ed. Association for the Conservation of Wildlife, Bangkok.
Macdonald, S.M., Mason, C.F. (1983). Some factors influencing the distribution of otters (Lutra lutra). Mammal Review, 13: 1-10.
Martin, D.J., Mcmillan, B.R., Erb, J.D., Gorman, T.A., Walsh, D.P. (2010). Diel activity patterns of river otters (Lontra canadensis) in south-eastern Minnesota. Journal of Mammalogy, 91(5): 1213-1224.
Mason, C.F., Macdonald, S.M. (1986). Otters, ecology and conservation. Cambridge University Press, Cambridge.
Melquist, W.E., Hornocker, M.G. (1983). Ecology of river otters in west central Idaho. Wildlife Monograph, 83: 1-60.
Mills M.G.L. (1989). The comparative behavioral ecology of hyenas: the importance of diet and food dispersion. In: Carnivore Behavior, Ecology and Evolution, Vol. 1 (ed. Gittleman JL), pp. 125-142. Cornell University Press, Ithaca, New York.
Nawab, A., Hussain, S.A. (2012a). Factors affecting the occurrence of smooth-coated otter in aquatic systems of the Upper Gangetic Plains, India. Aquatic conservation: Marine and freshwater ecosystems,
22: 616-625
Nawab, A., Hussain, S.A. (2012b). Prey selection by smooth-coated otter (Lutrogale perspicillata) in response to the variation in fish abundance in Upper Gangetic Plains, India. Mammalia,
76: 57–65.
Nolet, B.A., Kruuk, H.(1989). Grooming and resting of otters Lutra lutra in a marine habitat. Journal of Zoology, 218: 433-440.
Perrin, M.R., Carranza, I.D (2000). Activity Patterns of Spotted-Necked Otters in the Natal Drakensberg, South Africa. South African Journal of Wildlife Research,
30: 1-7.
Pocock, R.I. (1949). The Fauna of British India, including Ceylon and Burma. Volume 2, Mammals. Taylor and Francis, London.
Prater, S.H. (1971). The book of Indian animals. 3rd edn. Bombay Natural History Society, Bombay, India.
Schaller, G.B. (1972). The Serengeti lion. University of Chicago Press, Chicago.
Sollberger A. (1965). Biological rhythm research. Elsevier Publishing Company, Amsterdam.
Voigt, D.R., Tinline, R.L. (1979). Strategies for analyzing radio tracking data. In: Amlaner, C.J. Jr., MacDonald, D.W. (Eds.) A Handbook on Biotelemetry and Radio Tracking. Pergamon Press, Oxford and New York.
Résumé : Profil d’Activites, Comportements et Relations Interspecifiques de la Loutre à Peau Lisse (Lutrogale perspicillata) dans le Sanctuaire National de Chambal, Inde
Le profil d'activité, les comportements et les relations interspécifiques de la Loutre à poil lisse (Lutrogale perspicillata) avec les crocodiliens que sont le Gavial (Gavialis gangeticus) et le Crocodile des marais (Crocodylus palustris) ont été étudiés en suivant quatre loutres implantées de transmetteurs dans le Sanctuaire National de Chambal en Inde. A partir des pourcentages d’activités enregistrés, la tendance globale se présente sous forme d'une courbe bimodale dans laquelle apparaissent deux périodes de forte activité séparées par une période de relative inactivité. En été, la période de relative inactivité est supérieure à celle de l'hiver et de la mousson. Les loutres sont plus diurnes en hiver qu'en été ou à la mousson. Les déplacements et le toilettage sont les activités les plus courantes enregistrées sur 422 observations. Bien que les habitats et le régime alimentaire de la Loutre chevauchent ceux du Gavial et du Crocodile des marais, leurs activités diurnes sont différentes dans le temps et dans l’espace maintenant ainsi une cohabitation acceptable. Ceci permet d’éviter les conflits en assurant le partage des ressources et sécurisant siestes et toilettages.
Revenez au dessus
Resumen: Patrones de Actividad, Actividad Comportamental e Interacciones Interespecíficas de la Nutria Lisa (Lutrogale perspicillata) en el Santuario Nacional Chambal, India
Se examinaron los patrones de actividad, actividad comportamental e interacciones interespecíficas de la nutria lisa (Lutrogale perspicillata) con crocodílidos -el gharial Gavialis gangeticus y el cocodrilo de las marismas Crocodylus palustris; monitoreando cuatro nutrias radio-implantadas en el Santuario Nacional Chambal, India. En base al porcentaje de registros telemétricos activos, los patrones globales de actividad de las nutrias adoptaron la forma de una curva bimodal, en la cual dos períodos de alta actividad estuvieron separados por un período de relativa inactividad. En verano, el período de relativa inactividad fue mayor que en invierno y en los monzones. Las nutrias fueron más diurnas en invierno que en verano o en los monzones. Las actividades más prevalecientes registradas durante 422 avistajes, fueron el desplazamiento y el acicalamiento. Aunque los hábitats y la dieta de las nutrias se superpusieron con los del gharial y el cocodrilo de las marismas, su patrón diario de actividad fue diferente, y especialmente mantenían una estrategia mutuamente armoniosa, para evitar conflictos con recursos compartidos (para asoleamiento y acicalamiento).
Vuelva a la tapa